Abstract
Background
Asparaginase (ASNase) is one of the key components in the treatment of Acute Lymphoblastic Leukemia (ALL) and acts primarily by depletion of asparagine (Asn) from the blood. As lymphocytes and leukemic blasts are selectively dependent on the exogenous supply of Asn, exposure to this drug leads to a cessation of growth and the induction of cell death. Incomplete depletion of Asn either by poor penetrance of the drug (in the central nervous system) or support from the surrounding cellular microenvironment (in the bone marrow) may induce a cellular stress response that supports prolonged survival and prevents apoptosis, thereby enhancing the chance of relapses. Hence obtaining a detailed understanding of the cell intrinsic and extrinsic factors that contribute to a poor ASNase response is needed to further enhance the efficacy of this drug and reduce the number of relapses.
Method
To identify cell intrinsic determinants of sensitivity towards ASNase treatment, we performed a CRISPR/Cas9 based loss of function screen using NALM6 pre-B ALL cells exposed to a IC50 dose of ASNase. To facilitate rapid translation into clinical practice, we focused our screen on kinases as potential targets for pharmacological intervention. After treatment, genomic DNA from treated and untreated control cells was isolated, and incorporated gRNA sequences were amplified and sequenced (Illumina HiSeq) to identify gRNAs that were selectively enriched or depleted during treatment. The results were validated using targeted knockouts and by using small-molecule inhibitors. We used both ALL cell lines and a co-cultures of hTERT immortalized MSCs and ALL-xenografts for these functional validations.
Results
The anti-tumor effects of ASNase impinge on changes in cell metabolism that occur as a result of amino acid starvation. Consistent with this notion, we identified genes either directly involved in the amino acid response route (TRIB3) or inhibition of protein translation in response to amino acid starvation (GCN2). Indeed, knockout of GCN2 sensitized cells to ASNase treatment whereas depletion of TRIB3 was sufficient to render these cells more resistant to the effects of ASNase on cell growth.
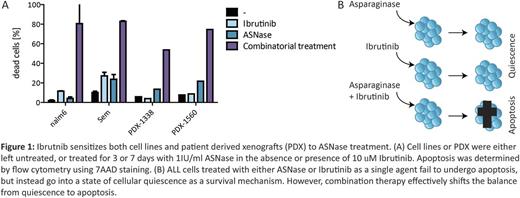
In addition, we found that Bruton Tyrosine Kinase (BTK), a hematopoietic-cell specific protein kinase acting downstream of the B-cell receptor, protects ALL cells from ASNase induced apoptosis. Indeed, targeted knockout as well as inhibition by the FDA-approved BTK inhibitor ibrutinib, strongly enhanced ASNase induced apoptosis in a variety of ALL cell lines (Figure 1). Moreover, we tested the effect of combination treatment in 35 different patient-derived xenografts samples, mostly representing high risk leukemia cases and covering a wide variety of ALL subtypes. In more than 75 % of the cases we observed synergy ranging from moderate to strong with a combination index (CI) <0.8.
Conclusions
These results show that
The nutrient stress response pathway is a key modulator of ASNase induced cell death
BTK provides a strong survival signal under (ASNase induced) nutrient stress.
The BTK inhibitor Ibrutinib is a promising candidate to potentiate ASNase therapy response in the patient
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract